 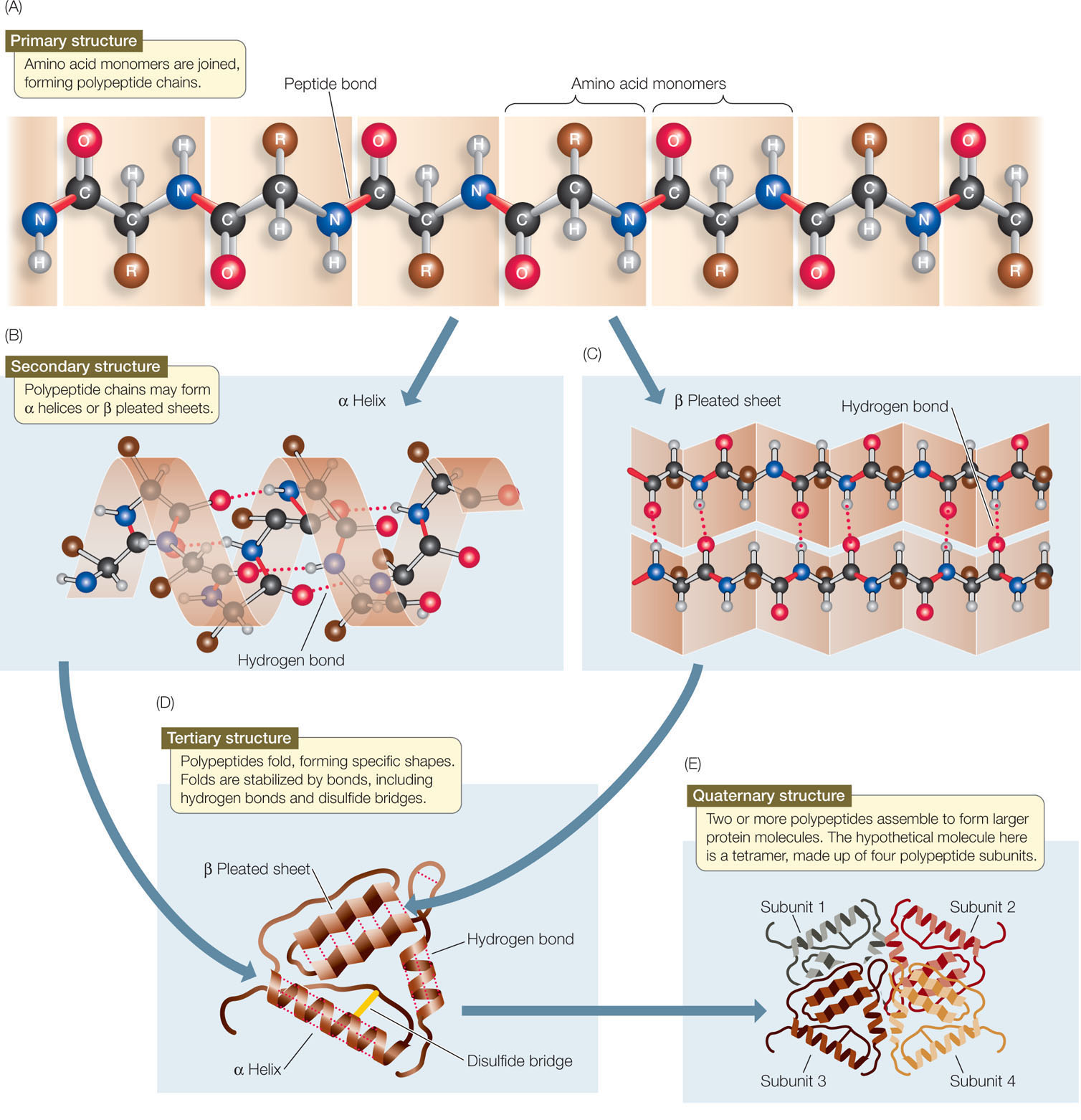
The other end is called the carboxyl terminus or C-terminus, since it contains the only free α-carboxyl group. The end of the protein that has the free α-amino group is referred to as the amino terminus or N-terminus. Synthesis of proteins occurs in the ribosomes and proceeds by joining the carboxyl terminus of the first amino acid to the amino terminus of the next one (Figure 2.19). 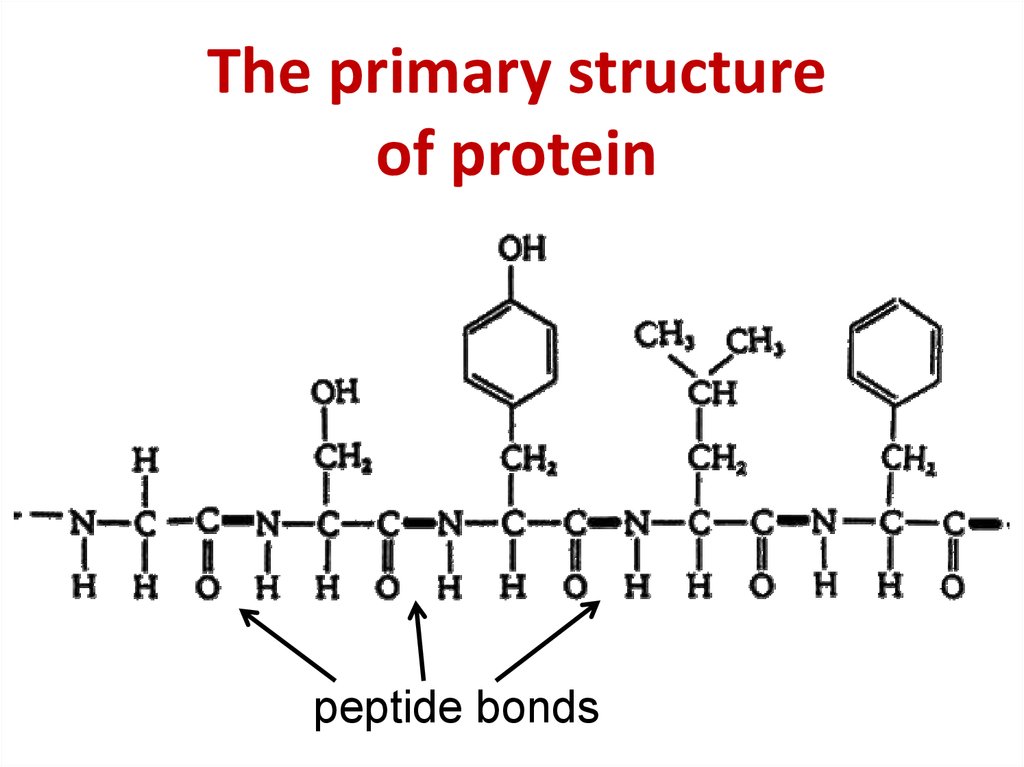
This is why mutations that alter amino acid sequence can affect the function of a protein. This structure, in turn, determines the protein’s function. The unique amino acid sequence of a protein is reflected in its unique folded structure. No two proteins with different amino acid sequences (primary structure) have identical overall structure. Sequence, structure and function Figure 2.19 Linking of amino acids through peptide bond formationĪs discussed earlier, the number of different amino acid sequences possible, even for short peptides, is very large. Hemoglobin, for example, undergoes an incredibly small structural change upon binding of one oxygen molecule, and that simple change causes the remainder of the protein to gain a considerably greater affinity for oxygen that the protein didn’t have before the structural change. Subtle changes Figure 2.18 - Sequence of a simple polypeptide WikipediaĮven very tiny, subtle changes in protein structure can give rise to big changes in the behavior of proteins. There are constraints on the conformations that proteins can adopt and these constraints govern the conformations that proteins display. However, proteins are not infinitely flexible. As we shall see, it is the flexibility of proteins that allows them to be amazing catalysts and allows them to adapt to, respond to, and pass on signals upon binding of other molecules or proteins. 
Proteins are flexible, not rigidly fixed in structure. For many proteins, it is not difficult to alter the structure. If you alter the structure, you alter the function - usually, but not always, this means you lose all function. Thus, when we use the term protein, we are usually referring to a functional, folded polypeptide or peptides. It is generally used, however, to refer to a folded, functional molecule that may have one or more subunits (made up of individual polypeptides). The term protein is sometimes used interchangeably with polypeptide, as in “protein synthesis”. It may or may not have folded into its final, functional form. We use the term polypeptide to refer to a single polymer of amino acids. Figure 2.17 - Four Levels of Protein StructureĪt this point, we should provide a couple of definitions. We shall examine protein structure at four distinct levels (Figure 2.17) - 1) how sequence of the amino acids in a protein (primary structure) gives identity and characteristics to a protein (Figure 2.18) 2) how local interactions between one part of the polypeptide backbone and another affect protein shape (secondary structure) 3) how the polypeptide chain of a protein can fold to allow amino acids to interact with each other that are not close in primary structure (tertiary structure) and 4) how different polypeptide chains interact with each other within a multi-subunit protein (quaternary structure). This conformation, in turn, will determine the function of the protein. The significance of the unique sequence, or order, of amino acids, known as the protein’s primary structure, is that it dictates the 3-D conformation the folded protein will have. Most proteins are much larger than this, making the possible number of proteins with unique amino acid sequences unimaginably huge. If we do the calculation for a short peptide of 10 amino acids, we arrive at an enormous 10,240,000,000,000 combinations. It becomes apparent that even a dipeptide made of just two amino acids joined together gives us 20 2 = 400 different combinations. 
To get a sense for the diversity of proteins that can be made using 20 different amino acids, consider that the number of different combinations possible with 20 amino acids is 20 n, where n=the number of amino acids in the chain. Proteins are made by linking together amino acids, with each protein having a characteristic and unique amino acid sequence. The versatility of proteins arises because of their varied structures. Life, as we know it, would not exist if there were no proteins. Among other things, protein enzymes catalyze the vast majority of cellular reactions, mediate signaling, give structure both to cells and to multicellular organisms, and exert control over the expression of genes. Virtually everything that goes on inside of cells happens as a result of the actions of proteins. The entire textbook is available for free from the authors at 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
